|
2/24/2024 0 Comments Ir spectroscopy organic chemistry
Hence, there is a net dipole moment resulting in a partial negative charge on oxygen and a partial positive charge on carbon. For example, in a C-O double bond (a carbonyl group), the electrons spend more time around the oxygen atom than the carbon atom because oxygen is more electronegative than carbon. If the electronegativities (the tendency to attract electrons) of the two atoms in a covalent bond are very different, a charge separation occurs that results in a dipole moment. When a molecule absorbs infrared light with a frequency that equals the natural vibrational frequency of a covalent bond, the energy from the radiation produces an increase in the amplitude of the bond vibration. 
Infrared light is electromagnetic radiation with wavelengths ranging from 700 nm to 1 mm, which is consistent with the relative bond strengths. Double and triple bonds can be considered as stronger springs, so a C-O double bond has a higher stretching frequency than a C-O single bond. Thus, C-H, N-H, and O-H bonds have higher stretching frequencies than C-C and C-O bonds, as hydrogen is a light atom. The frequency is typically measured in wavenumbers, which are expressed in inverse centimeters (cm -1).įrom Equation 1, the frequency is proportional to the strength of the spring and inversely proportional to the masses of the objects. This frequency is given by Equation 1, where k is the force constant of the spring, c is the speed of light, and µ is the reduced mass ( Equation 2). Naturally, this bond stretches and compresses with a certain vibrational frequency. 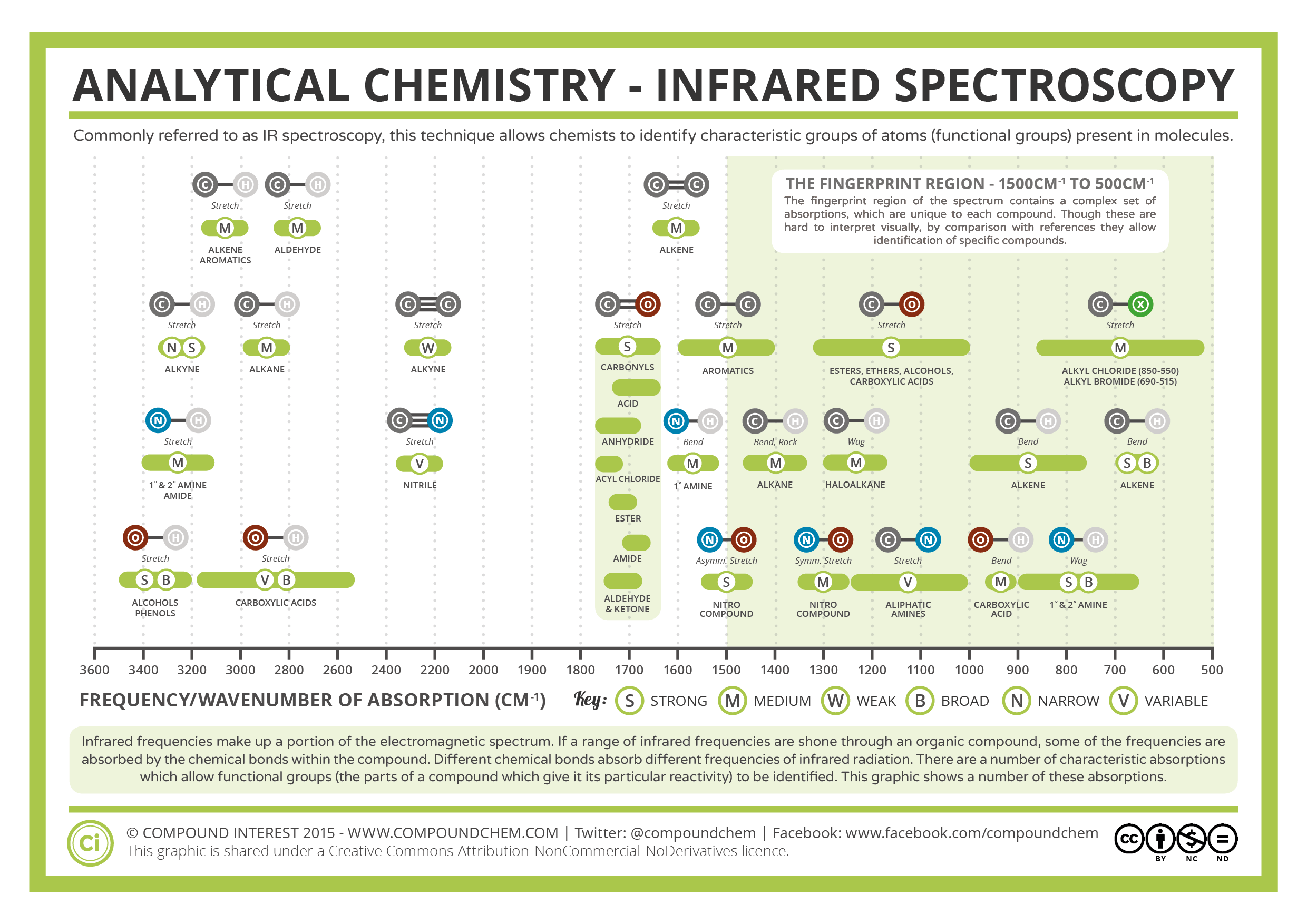
A covalent bond between two atoms can be thought of as two objects with masses m 1 and m 2 that are connected with a spring.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
